ADVANCED MICROCURRENT SCIENCE FOR ANTI-AGING
Dynamic Intelligent Face and Body Sculptors
Professional non-invasive facial and body sculptors that feature next generation microcurrent technology. Take your spa offerings to the next level with revolutionary advancements that are cleared by the FDA and made in the USA.
Dynamic Microcurrent Facial & Body Systems for Estheticians, Salons, Med Spas & More
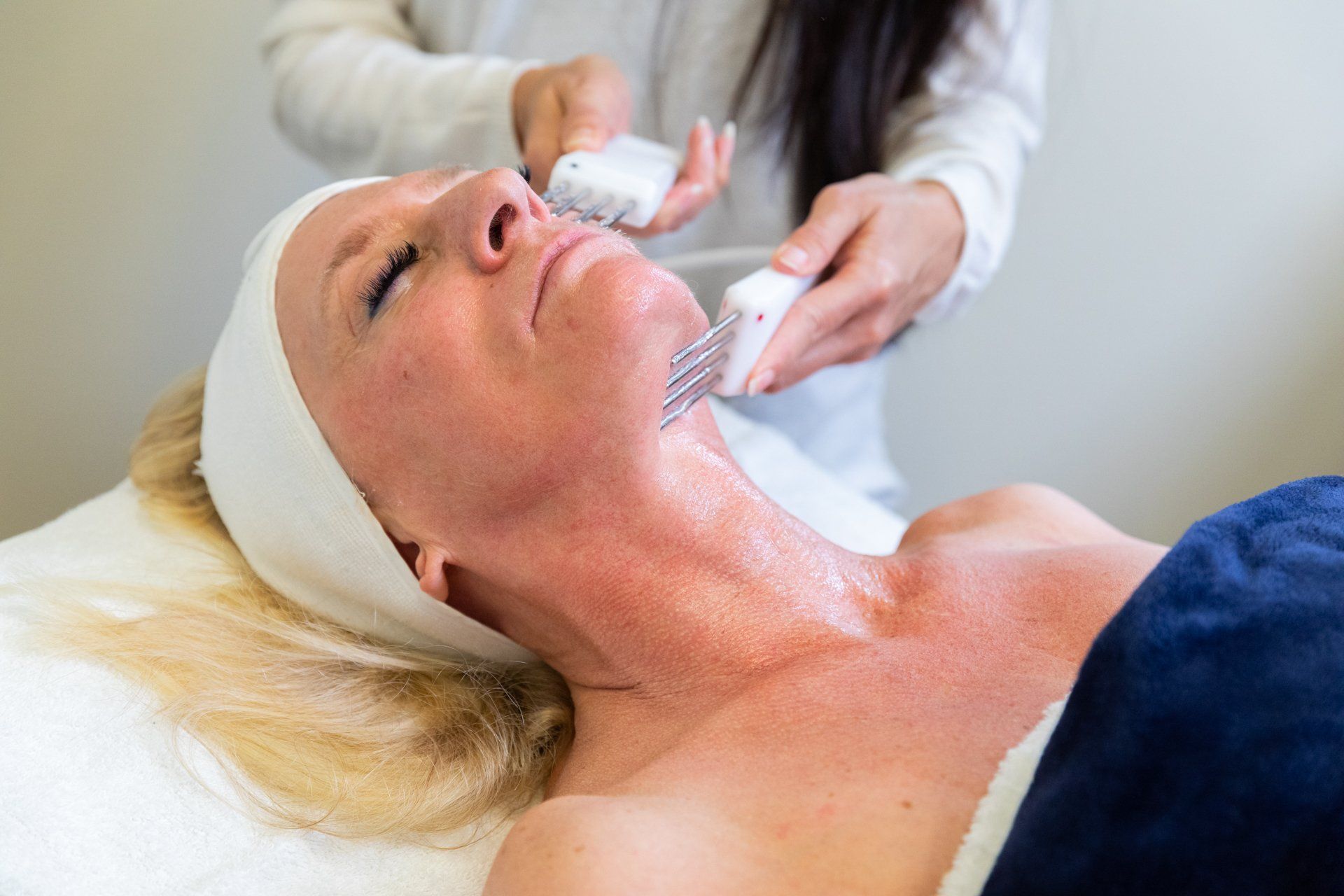
NeurotriS introduces the only Absolute True Dynamic Intelligent Microcurrent facial & Body Sculptors on the market that features a Bio-feedback monitoring loop optimizing results based on an individual’s skin impedance at the time of treatment.
Our latest third Generation Dynamic Machines integrate manual and automatic program modes. In Automatic mode, the machine will change the signal several times throughout the treatment session while maximizing signal integrity based on skin density and type for maximum results. Exclusive Patented Technology to NeurotriS.
Our Products
-
The NEW SVX650
ButtonThe newest addition to our products is one you won't want to miss out on, results are seen quickly, and clients leaving feeling better than ever. Experience the life changing technology now offered by NeurotriS. Learn More
-
SX-2500
ButtonThe NeurotriS PicoWave is a Dynamic Intelligent System that features a Bio-Feedback monitoring loop that automatically sequences different signals throughout the duration of the preset program optimizing results based on an individual’s skin type at the time of the treatment. Learn More
-
SX-3800
ButtonThe Sx-3800 is our top facial toning microcurrent system that uses our patented Constant Waveform Morphology to monitor skin impedance across 4 independent channel outputs. Learn More
-
SX-4500
ButtonThe Sx-4500 is a Level I & Level II facial & body toning microcurrent system that uses our patented Constant Waveform Morphology to monitor skin impedance across 16 independent body and 2 independent face channel outputs. Learn More
-
PICO Toner
ButtonThe PICO Toner is a single output personal microcurrent system that uses our patented Platinum Nano Technology featuring 2 preset programs and a long battery life. Learn More
-
AMNA Lift
ButtonThe Lift Probe adds the power of Dynamic Microcurrent to Jane Mann’s ANMA facial contouring tool that uses specialized facial massage techniques to help restore definition, collagen, and elastin to the skin. Learn More
Accessories
All the SX-Series microcurrent accessories you need to go with the products you love. Learn More
Celebrities love the power of NeurotriS
Paris Hilton

Madonna

Paula Abdul

Jennifer Aniston

Want to know more about "The Madonna Facial?"
Get to know Dynamic Intelligent Microcurrent and why to choose NeurotriS! Barry did a great interview with Cherie Callahan who explains what it is and how it works.
FDA 510K Clearance, Licenses & Certificates
A-1 Engineering's NEW Irvine facility has been recently inspected by the FDA & CDPH and was found to be in full compliance to the FDA Quality System Regulation Title 22 Part 820 quality management system & procedures, device history records (DHR), safety & performance. NeurotriS / A-1 Engineering is an approved licensed facility that meets applicable FDA requirements and specifications for current good manufacturing practices (CGMP’s) license # 78634. NeurotriS / A-1 Engineering is the only USA Licensed approved Microcurrent device manufacturing facility approved to sell and distribute worldwide.
NeurotriS / A-1 Engineering is the only U.S. manufacture Licensed by the Federal and state regulatory bodies.
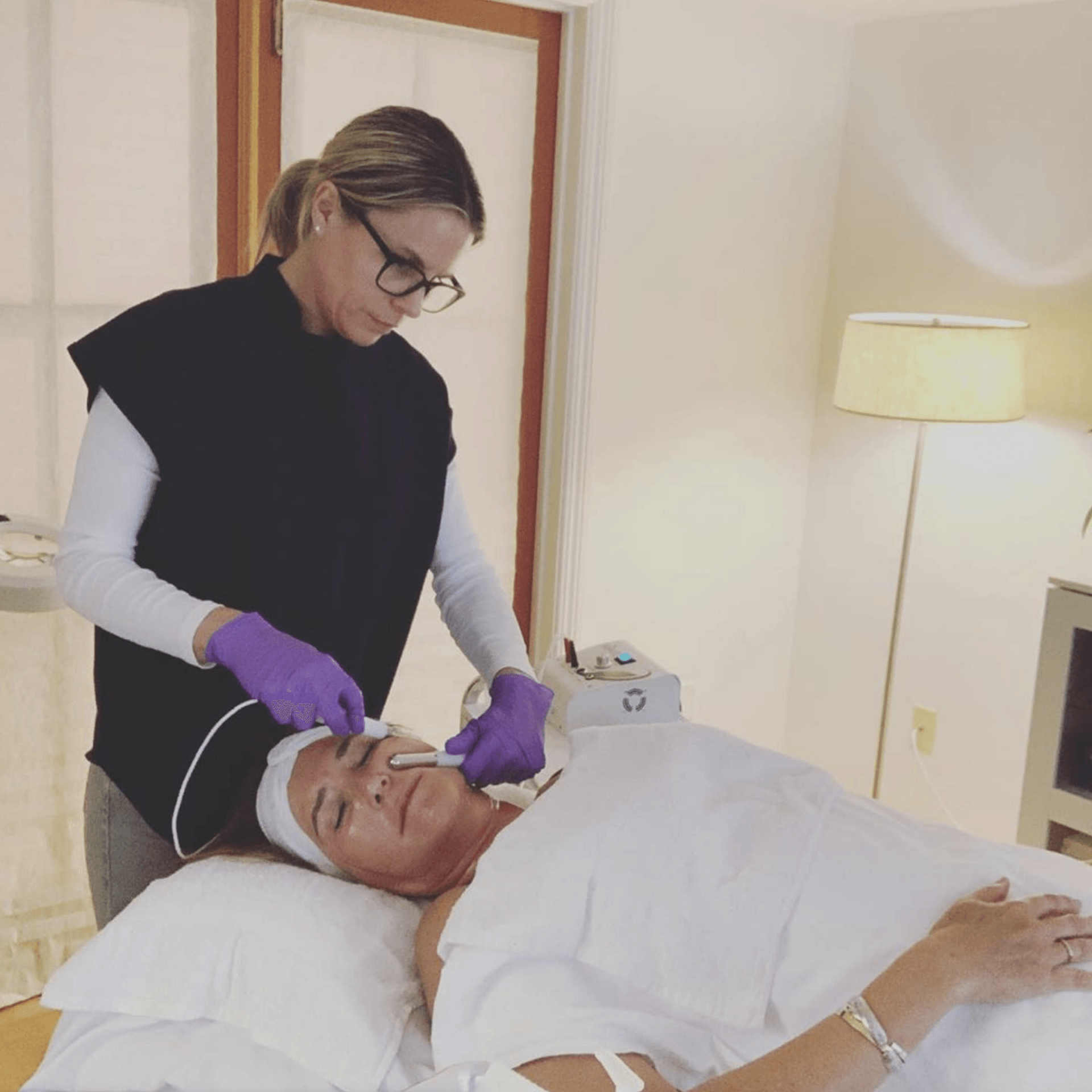
Personal Face & Body Toner
NeurotriS introduces the worlds most advanced personal face & body toner. The NeurotriS PICO Toner™ is the only hand held microcurrent device intended for home use that uses personal Silver Gloves™ to bring back the natural "glow" of youthful hues for a more youthful appearance. The patented NeurotriS Silver Gloves™ are designed to deliver the specific preset signatures evenly allowing for more improved results!
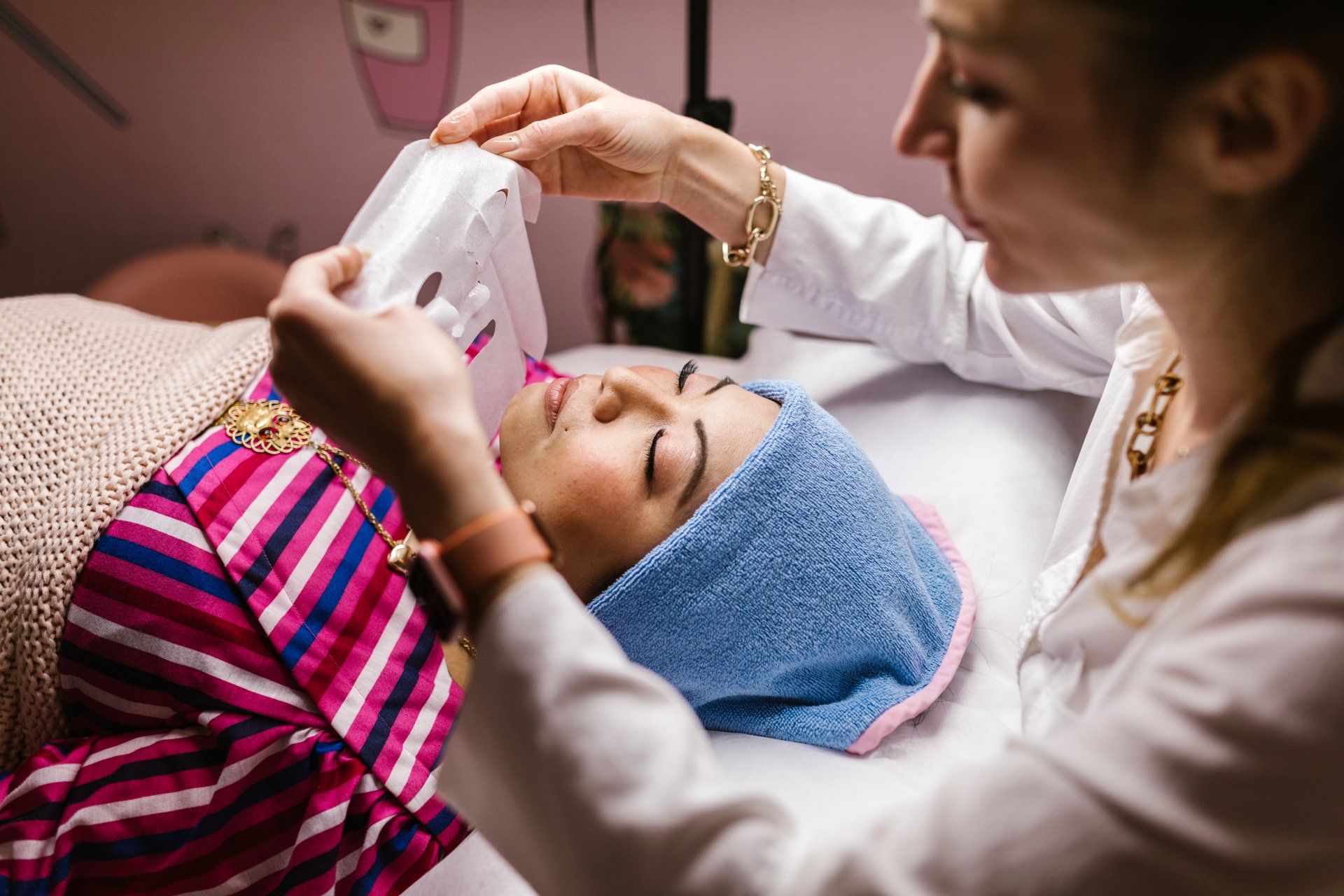
NEW Dynamic Facial System
NeurotriS Dynamic Intelligent Systems features a Bio-Feedback monitoring loop that automatically sequences different signals throughout the duration of the preset program optimizing results based on an individual’s skin type at the time of the treatment.
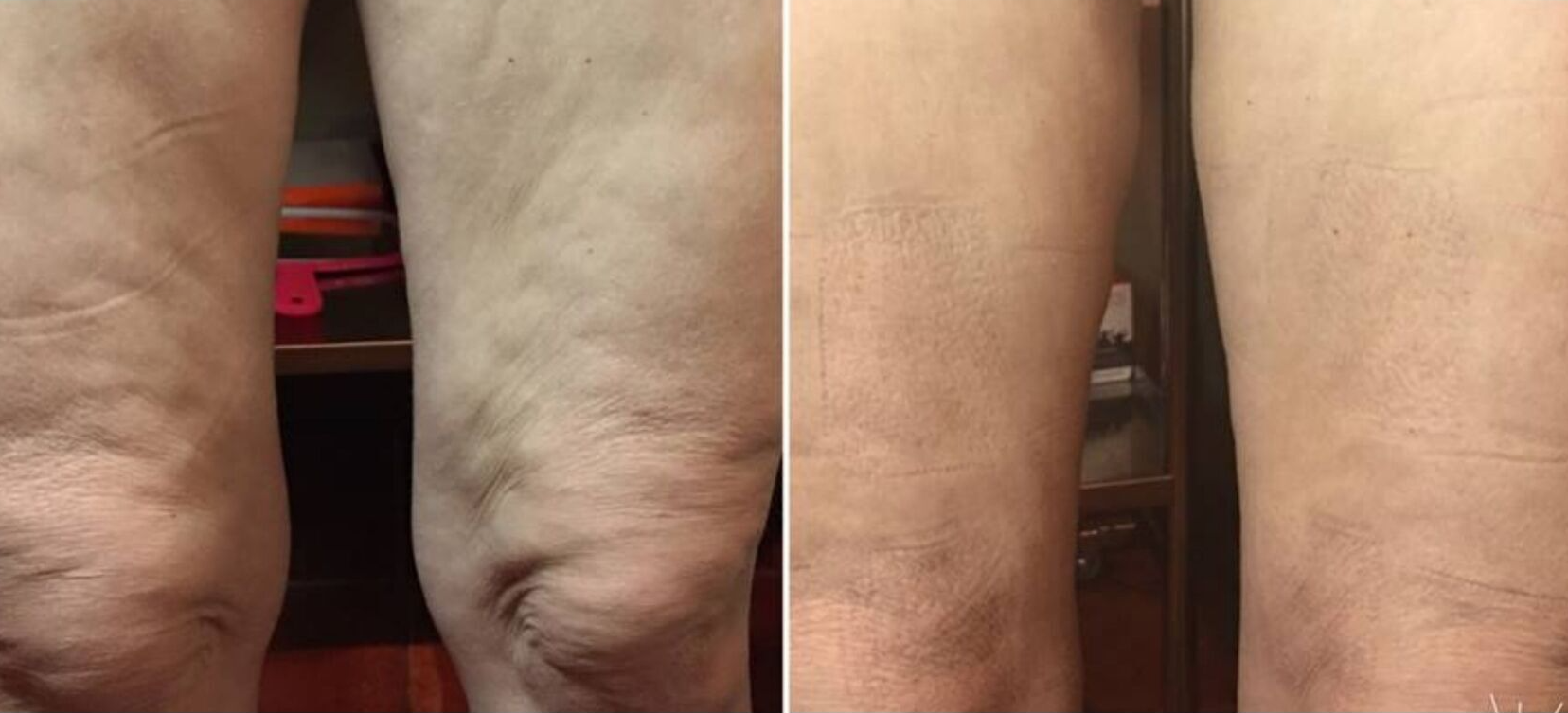
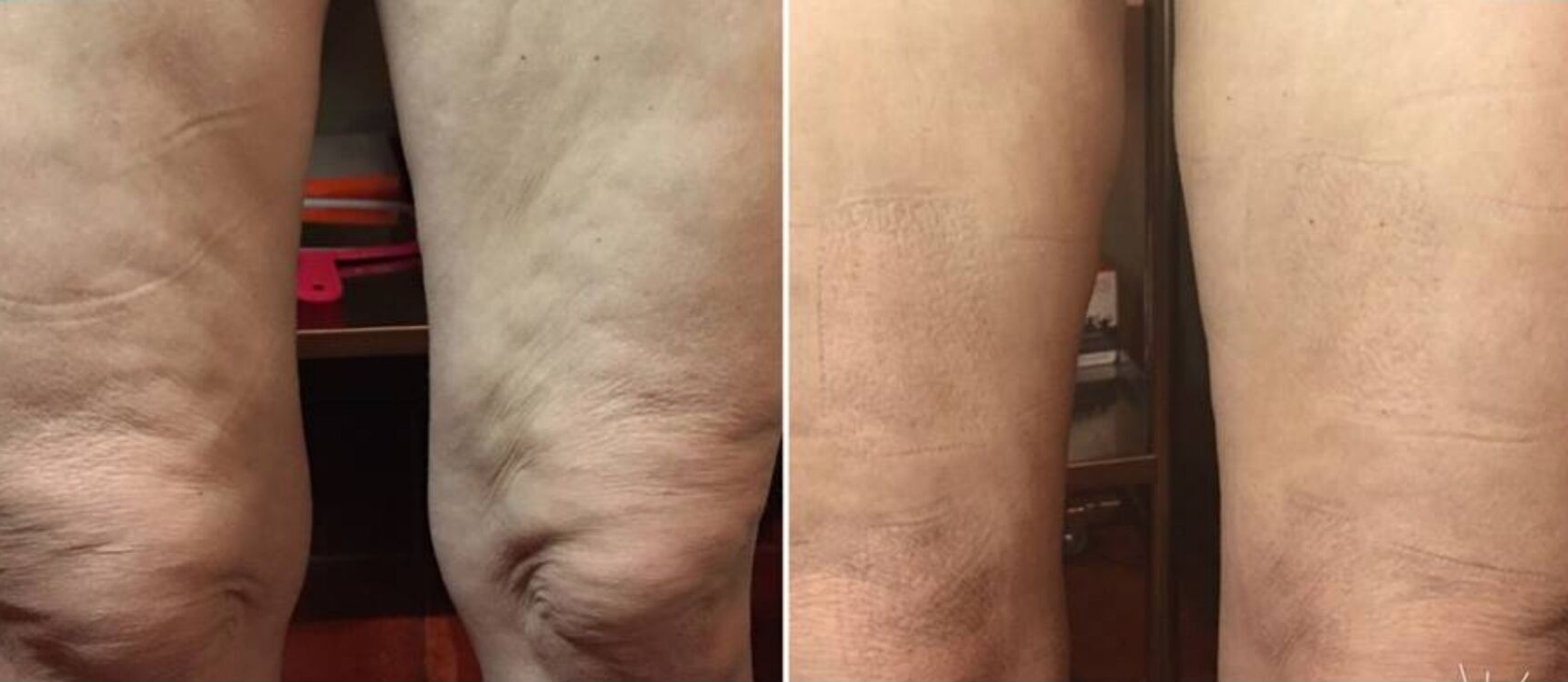
Dynamic Body System
This process uses Specific Frequency Signatures to give you instant visible results. See an immediate reduction in your dress size! Experience a relaxing session from the NeurotriS microcurrent machine that Vogue Magazine calls, "The Miracle Machine".
Take a Look at our Blog Post
Your Success is our Goal
Neurotris is a great place! Enjoy the treatments and results. My waistline and inner thighs were my target areas where inches and fat melted away! Beats those invasive procedures out there. Also strength training my back and core. Patricia works with you and makes sure you are comfortable in the best of care.